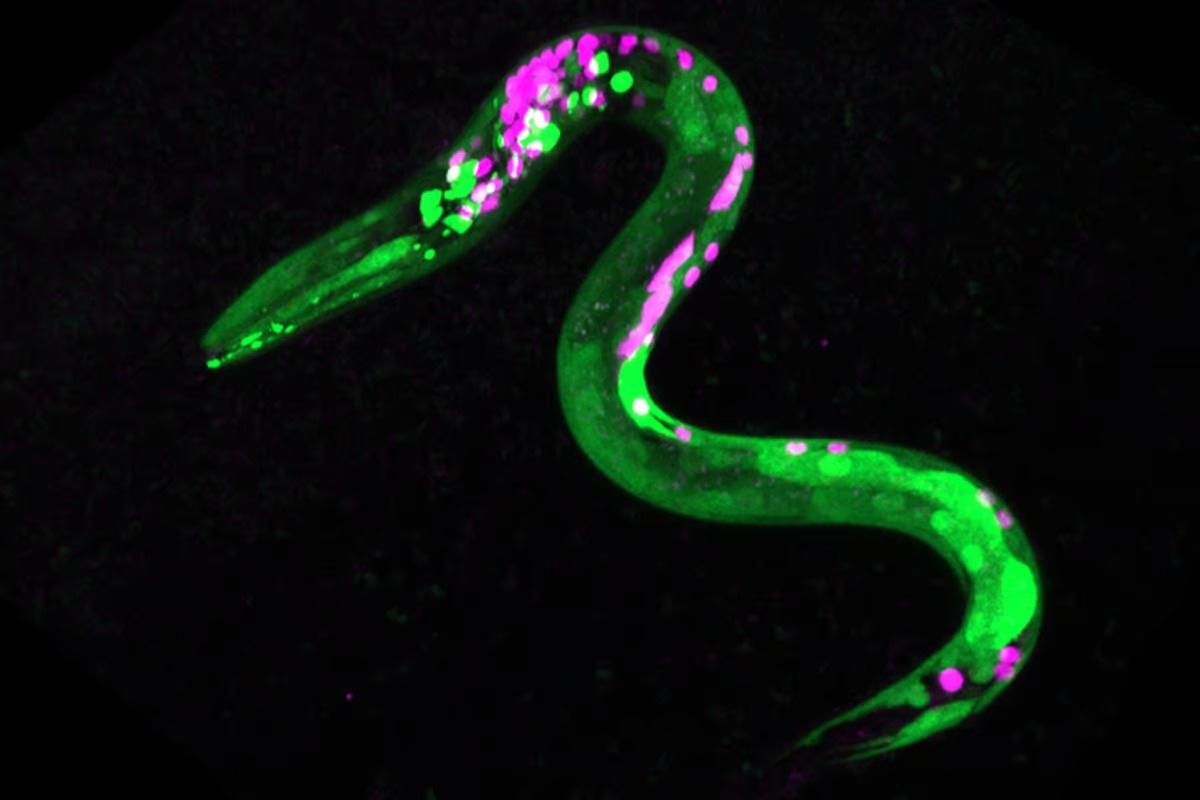
 Image of a C. elegans worm genetically engineered to show fluorescence – the green dots are neurons that have responded to cannabinoids. Stacy Levichev/University of Oregon (CC BY-SA)
Image of a C. elegans worm genetically engineered to show fluorescence – the green dots are neurons that have responded to cannabinoids. Stacy Levichev/University of Oregon (CC BY-SA)
–
The phenomenon brought on by cannabis use and known colloquially as ‘the munchies’ has been known about for centuries, although scientists have a fancy term for it: hedonic amplification of eating.
Hedonism is the ethical theory that proposes that pleasure represents the highest good and that the proper aim of human life is to seek pleasure and self-indulgence. It follows, then, the hedonic amplification of eating that follows the smoking or ingestion of cannabis refers to a heightened desire to eat foods that are pleasurable.
Cannabis is a cannabinoid. The body creates endocannabinoids, similar to cannabinoids, as part of a signaling system responsible for maintaining bodily homeostasis or balance. A key endocannabinoid is anandamide, which binds to the same brain receptors as delta-tetrahydrocannabinol (THC), the main psychoactive component in cannabis.
Animal studies have found a strong link between endocannabinoid signaling and energy homeostasis; that is, matching caloric intake with energy expenditure. These studies have also shown that administering THC or endocannabinoids not only increases food consumption, but it also changes food preferences in favor of calorically dense foods like those that are high in sugar or fat.
Researchers from the University of Oregon gave cannabis-like substances to tiny bacteria-eating worms to see if their food preferences changed.
Caenorhabditis elegans, or C. elegans for short, are biology’s favorite worms because they share many genes with humans, and many of the molecular signals that control the worm’s development are also found in humans.
The researchers soaked the worms in anandamide and put them in a T-shaped maze. On the left side of the maze was high-quality food; on the right, low-quality. From an evolutionary standpoint, “high-quality” food means food packed with calories that ensure survival. So, the worm equivalent of junk food.
“The endocannabinoid system helps make sure that an animal that’s starving goes for high fat and sugar content food,” said Shawn Lockery, corresponding author of the study.
While the worms naturally exhibit a preference for high-quality food, the researchers found that in anandamide-affected worms, the preference was much stronger. The worms flocked to the left side of the T-maze and stayed there for longer periods than usual. From their results, the researchers concluded that anandamide induces hedonic feeding in C. elegans.
“We suggest that this increase in existing preference is analogous to eating more of the foods you would crave anyway,” said Shawn Lockery, corresponding author of the study. “It’s like choosing pizza versus oatmeal.”
The researchers also conducted experiments to see how anandamide affected the worm’s neurons. Using C. elegans that had been genetically engineered so that certain neurons and muscles became fluorescent, they found that the worms affected by anandamide were more sensitive to the smell of high-quality food and less sensitive to the smell of low-quality food.
But how does this study relate to humans? The researchers say it shows how long the endocannabinoid system has been around. Humans and worms last shared a common ancestor more than 600 million years ago, and yet, today, both species exhibit similar food preference changes in response to cannabinoids.
“It’s a really beautiful example of what the endocannabinoid system was probably for at the beginning,” Lockery said.
The study’s findings also suggest that C. elegans could provide a useful model for future studies of the endocannabinoid system and the development of drugs that target cannabinoid receptors in the body.
Currently, medicinal cannabis is used to treat several medical conditions, but it has side effects. For example, while it might effectively reduce pain, it can negatively affect concentration.
“The ability to rapidly find signaling pathways in the worm could help identify better drug targets, with fewer side effects,” said Lockery.
The study was published in the journal Current Biology.
Source: University of Oregon via EurekAlert!
–